Biological dentistry

by Dr. Dominik Nischwitz
The dental contribution to chronic disease – endotoxins 24/7.
The immune system is designed to repel bacteria, fungi, viruses and other microorganisms. In Western countries/industrialized nations there is currently an epidemic of chronic systemic diseases [1,2]. There are many causes, but hyperreactivity of the innate immune system plays a crucial role in this equation.
Apart from classic, technically characterized dentistry, in biological dentistry all scientific basic rules are also used. As the saying goes: “The mouth is a mirror of health”: the basic principles of chemistry, physics and biochemistry are examined more closely under the magnifying glass and a connection is made between today’s common chronic-inflammatory systemic diseases.
Various, partly even toxic metals (mercury/amalgam) are used routinely because they work well and are durable. Dead and endodontically treated teeth are found in the oral cavity of the average adult. Inflammations at the root apex are common – they are dismissed as harmless or simply removed symptomatically with surgery. At least the oral microorganisms are kept in check in some practices with consistent prophylactic cleaning. Since the human body and its strong immune system can compensate relatively well and conventional medicine does not consider the body to be an integrative system, focusing much more on its individual parts, the link between the oral cavity and symptoms elsewhere in the body has not been well established.
The mouth as construction site in the body
Teeth are exactly like the liver, stomach or intestine as well as organs with their own blood and nerve supply, and literally in this case, have a cranial nerve with an autonomous nerve system [3]. They are the organs that are anatomically closest to the brain.
Widely varied, at times highly toxic materials are routinely embedded in this sensitive area under the guise of technical durability – with often serious consequences for the entire body.
Most interference fields in the body are in the oral cavity. Apart from classic problems such as caries and periodontitis, these include:
Metals – inherited burdens from the last century
Picture: Old amalgam fillings and metal pins.
For different metals such as mercury, gold, platinum, copper, cobalt, aluminum, iron and chrome, the cytotoxic (e.g. neurological diseases), immunological (autoimmune disorders) and mutagenic (e.g. cancer) effects as well as effects on the metabolism (e.g. oxidative stress) are scientifically well documented [4–13]. Metal components can usually be detected throughout the entire body a few days after installation in the mouth.
One must always differentiate between immunological and toxicological problems. While practically every metal represents a foreign body for the immune system and exhibits allergenic potential, amalgam in particular plays a crucial role from a toxicological point of view.
Amalgam – highly toxic hazardous waste in our body.
Even today amalgam is routinely used in most dental practices. On the one hand this is because it is a material that is easily processed and holds for a long time, and on the other because it is subsidized by health insurance companies and is therefore free.
In Norway (2008) and Sweden (2009) mercury has been completely banned for some time, which of course also includes fillings. In Russia amalgam was already abolished completely at the end of the 1970s.
In contrast, the Federal Association of Dentists (BZÄK) requires amalgam as filling material.
In practice, amalgam must be disposed of after removal as a highly toxic hazardous waste – this fact alone should give pause for thought. Amalgam consists of 50% mercury (Hg), which contrary to what is often assumed is not firmly set in the filling after mixing.
Picture: Example composition of an amalgam filling: 51% mercury (Hg), 21.5 % silver (Ag), 14.8 % tin (Sn) and 12.7 % copper (Cu) (according to manufacturer’s data).
Chewing, grinding, tooth brushing and hot or cold beverages release a certain amount of mercury vapor every day. All this takes place within the microgram range; however, if one considers that even one molecule of Hg can destroy nerve cells, this cannot be underestimated. A study by Leong and Lorscheider showed that inorganic Hg quantities of 0.02 ng Hg/g led to a complete destruction of the intracellular microtubules and to a degeneration of nerve axons [14]. Hg is considered the most toxic non-radioactive element and thus exceeds all other known elements, such as lead, cadmium and arsenic, in some cases many times over [15–17]. In animal studies, after 14 days of amalgam wearing time, pathological changes could already be detected in the brain [18,19].
Picture: Smooking Tooth – www.uninformedconsent.org: Hg vapor from a 50-year-old filling – shown under fluorescent light
About 2–3µg mercury vapor is released daily per filling and that is over an average wearing time of 20 years. This therefore qualifies as low-dose, chronic poisoning. In numerous studies an approximately 2–5-fold increase in mercury in the blood and urine was observed in living amalgam carriers; investigations on deceased patients found even 2–12-fold increases in Hg levels in different body tissues. According to these studies, amalgam is the main source for mercury load in the human body [8,20–39]. Mercury can trigger any symptom and is not tolerable in the body for these reasons. The human body is extremely intelligent and stores, wherever possible, fat-soluble toxins in the metabolically inactive connective or adipose tissues. However, in athletic people or those with a low percentage of body fat, the toxins are frequently deposited in the nerve tissue or brain. In particular, babies who are nursing or even during pregnancy in the mother’s body are at risk, as Hg can penetrate the placenta. The amount of mercury in breast milk and amniotic fluid clearly correlates with the number of amalgam fillings the mother has [40–51]. Since amalgam fillings are the main source for poisoning with mercury and other heavy metals, these should be removed either in the event of chronic illness or for preventive reasons.
However, it is exactly with this removal that most mistakes are made. Usually the dentist simply drills the filling out with no safeguards because he does not know about the problem specified above (not university doctrine). Nevertheless, a very large amount of highly toxic, inorganic mercury vapor (Hg0) develops; often patients react after even a routine amalgam removal with neurological complaints, chronic fatigue, joint and muscle complaints or other recently acquired symptoms.
For this reason the removal of the fillings under absolute safeguards is indispensable.
Picture: Chlorella
Since the body stores mercury during wearing time, particularly in the brain with a half-life from several years to decades, [52-59], it makes sense to support the body after metal removal with an active heavy metal decontamination [60–64]. The brain serves here as a type of waste disposal – once in the cell, Hg vapor very quickly oxidizes through catalases to Hg2+ and can thus be covalently bound to the thiol group of proteins, which in turn changes or even blocks biological activity. In the gastrointestinal tract the Hg from amalgam is converted from microorganisms into the organic, methylated form [65–67]. Leistevuo et al. were able to prove a 2 to 3-fold increase in organic methylmercury in the saliva of amalgam carriers compared to subjects without amalgam. Fish consumption was identical in the two groups in both quantity consumed and frequency [66]. Methylated mercury in the gastrointestinal tract seems to be much more toxic than methylmercury from fish consumption, since the Hg in the fish is already bound to amino acids, whereas it is directly formed in the body and is thus is clearly more reactive [68].
A package program cannot, as can be read on the Internet, simply be implemented for heavy metal detoxification – rather these so-called detox programs can only lead to problems, because not every patient is able to simply secrete heavy metals or other toxic substances. Knowledge of the patient’s individual biochemistry as well as of possibly existing micronutrient deficits are particularly necessary for already existing health problems. In order to be able to deal with the body’s decontamination reactions, one should put themselves in the hands of experienced physicians or naturopaths.
A complete decontamination can be correctly undertaken only after the consistent removal of all interference fields in the oral cavity. This does not include only the known amalgam.
Synergy of inflammation and metals
Through the metabolism of bacteria, sulfurous protein decomposition products such as hydrogen sulfide (H2S), thioethers and mercaptans develop, which have a high bonding capacity to heavy metals [69–71]. These result in organic metal compounds of substantially increased toxicity, for example dimethylmercury [65–67] – e.g. amalgam filling on an endodontically treated tooth. Chemist Karen Wetterhan used this material in the laboratory to examine the disease-related dynamics of molecules at the cellular level. A drop of dimethylmercury fell on her latex-gloved hand and within a few hours spread through her skin to her entire body. Despite subsequent chelation therapy, she died few months later of mercury poisoning. The concentration of these poisonous molecules is many times lower through the metals in the mouth; however, it is important to know that we as dentists are exposing patients and ourselves [34] to these highly toxic substances and installing these as a source 24 hours a day, 7 days a week for the entire wearing time in the body.
Picture: Mercury vapor from amalgam fillings can mutate in conjunction with bacterial biotoxins into highly poisonous supertoxins (dimethylmercury).
The battery effect
A battery develops when two different metals are brought into a conductive solution. The base metal ions go toward the electrochemical series in a solution and flow toward the nobler metal, electrons are released – a current flows. The saliva is the optimal electrolytic solution due to high mineral content. A classic example is a gold crown beside an amalgam filling or a gold abutment on a titanium implant. This is referred to in this case as a galvanic current or the battery effect.
These comparatively high dental mouth currents lead to the corrosion of the metals in the course of wearing time, which inevitably correlates with the problems of metal toxicity.
In addition, patients’ increasing electrical sensitivity is a result of the exponentially increasing diffusion of microwaves by WI-FI and cell phone emissions.
It must be emphasized that metals in the body act like small antennas that can disturb the sensitive action potential of the cell completely. Areas of tension develop, which sensitively disrupt the central nervous system. Inevitably one is exposed to electrical smog everywhere [72]. The standard absorption rate of electromagnetic fields can be increased 400 to 700-fold through the use of a cell phone (ringing or SMS reception) in combination with metals in the mouth [73].
Electrogalvanism and the resulting electrical sensitivity can frequently be the cause of a lack of concentration and memory loss, sleeplessness, non-specific symptoms such as a sharp pain or pressure in the chest, unexplained tachycardia, tinnitus and hearing loss, etc. [74].
Picture: Classic scenario: Gold crown next to amalgam filling – the battery effect.
Interference fields in the oral cavity
Endodontically treated teeth
This topic is in the author’s opinion a highly significant chapter in the history of dentistry and therefore in his article “Root to Disease” it was considered in greater detail [75]. Endodontically treated teeth represent chronic-inflammatory sites that can lead to chronic problems both locally but more often in other areas of the body. More than 100 years ago Weston Price already coined the term focal infection for this purpose.
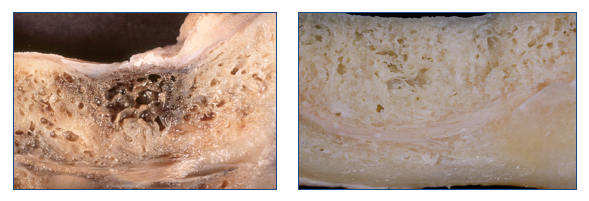
Without a blood, nerve and lymph supply the tooth is only dead organic tissue without function, which thanks to its anatomy represents the perfect cavity for pathogenic microorganisms. There are between 30 and 75,000 dentin canals per mm2. If all the dentin canals of a root were lined up, they would cover a distance of approx. one kilometer. Pathogenic bacteria exist in this widely ramified canal system of a root and form highly toxic sulfur compounds (thioethers, mercaptans) [69–71], which for their part can block vital enzymes at their active center. As nonliving organic tissue can begin to decompose over time, necrogenic material with increased toxicity (putrescine and cadaverine) can also develop.
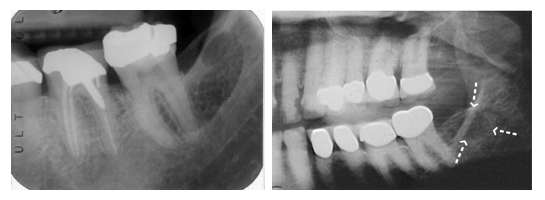
Picture: Endodontically treated teeth on an x-ray and in real life. Dead tissue leaves traces
Inflammations at the root apex
The nonspecific immune system reacts to this infectious site with the increased production of proinflammatory cytokines (TNF-α, IL-1, INFγ). This subclinical activation of the tissue macrophages leads to chronic inflammation of the surrounding tissue and to a widening of the periodontal space and to possible cyst formation. Apart from the toxicity of sulfur-hydrogen compounds (thioethers/mercaptans), it is also common for a patient to have an allergic reaction to these substances. Root filling materials are also problematic and usually contain classic allergens such as epoxy resin, Peru balsam or rosin. Problems do not usually show up directly on site, but systemically, somewhere else in the body. Therefore, they are not always easy to diagnose.
The teeth belong to the most important subsystems within a network of self-regulating sub-ranges in the body. Teeth and their periodontium (=odonton) have a relationship with other physical structures and organs. Reinhold Voll coined the term odonton and identified the direct and close interrelations between individual odontons and the different areas of the body. Interactions and positive and negative influences in terms of a remote effect are possible in both directions:
A disturbed organ can pathologically affect the associated odonton and conversely a sick tooth or its periodontium can disturb its correlating organ. Classic interference fields next to endodontically treated teeth are displaced teeth and wisdom teeth, non-vital teeth, metal fragments and other foreign bodies, cysts and chronic inflammations in the jawbone.
Chronic inflammations in the jawbone (residual osteitis/NICO)
Undetected by conventional x-ray, chronic inflammations in the jawbones often occur, usually resulting from old, not optimally healed tooth extraction wounds, dental germs or foreign bodies [76]. Here, similarly to endodontically treated teeth, toxic substances and inflammatory mediators (TNF-α, IL-1, RANTES) develop, which can cause various symptoms in other parts of the body – neurological (NICO) or joint problems are particularly frequent. These can be well diagnosed on suspicion when using 3D digital volume tomography (DVT).
The therapy consists of the complete surgical removal of the areas altered by inflammatory activity followed by disinfection with ozone. In the author’s practice insertion of a PRGF® membrane (Platelet Rich in Growth Factors) obtained from autologous blood has proven successful. The patient’s freshly withdrawn venous blood is centrifuged for approximately 8 minutes and thus activated. After 30 minutes at body temperature, the membrane is ready to insert. PRGF® Technology is of 100% autologous origin and therefore fully biocompatible.
Picture: J Oral Pathol Med 1999; 28:423.
Bite problems and dysfunctions of the mandibular joint
The bite is the thermostat for the body’s structural component. The chewing process is primarily needed for our survival. In the last 100 years, in addition to iatrogenic and traumatic occlusion problems, innate and developmental abnormalities of the upper and lower jaws have become an everyday occurrence. This seems to have primarily epigenetic causes, notably nutrition plays a crucial role here. Food, mainly sugar and white flour products, seems to be directly linked to the development of malpositions of the teeth and jaws [77]. Nowadays, it has become relatively rare in industrialized nations for a child or adolescent not to have some type of orthodontic device.
Tooth and eye level are directly connected to one another: in every mammal the eyes are fixed on the horizon (labyrinthine righting reflex). Even the slightest changes in occlusion in the micrometer range lead to an imbalance in this structure. The compensation occurs through simple control mechanisms in the muscular and ligamentous apparatus, but initially through a shift of the cranium along the sutures and meninges. This mainly manifests itself in tension of the deep neck musculature; however, over a longer period it is transferred on to the shoulder and pelvic girdle. Thus, in the long-term misalignments and malpositions can develop.
The tension also leads to a reduced blood supply to the brain in certain areas as well as decreased blood flow and lymphatic drainage from the brain. Even a millimeter loss at bite level leads to a clear loss in lymphatic drainage and concomitantly to an accumulation of toxins in the brain area. Apart from the usual symptoms of craniomandibular dysfunction and myoarthropathy, a myriad of other symptoms can arise as a result, including concentration and memory loss (reduced supply of the neurotransmitter acetyl choline due to deficient blood circulation), migraine, sleeplessness, neck pain, pain in the lower back area, right up to depression [74].
The mouth could be compared to a large building site that is never finished. Every day the body must expend workers and building material in terms of nutrients and ATP in order to compensate for this building site.
24 hours a day, 7 days a week nutrients are used for the body’s ability to compensate, in other places in the body deficits or even deficiency symptoms occur. The toxic substances are increased and the body can no longer take care of the disposal – the barrel overflows. Here the key partly lies between biological dentistry and today’s common chronic illnesses. According to Straub et al., the body’s energy loss due to a chronically activated immune system is calculated at roughly 30 percent [78]. These patients are also missing a third of the energy in the morning after they get up. Small wonder then that chronic fatigue has become a widespread disease. Additionally, the body’s individual, genetic detoxification activity naturally plays an important and crucial role in this equation. This often includes inadequate nutrition, food intolerances, chronic bacterial and viral infections as well as other environmental influences (plastics, pesticides, solvents, insecticides, preservatives, etc.), to which the population is exposed daily. As such, consistent biological cleaning of the oral cavity plays an important role not only for chronically ill patients but also, if not more so, for everyone in terms of prevention.
The Alternatives
It is generally known that highly toxic mercury vapor (Hg0) develops with the removal of amalgam. Therefore, for the removal of the metals, in particular amalgam removal, maximum safeguards must be in place to protect the patient, but also above all for the dentist and his team who come into contact with the vapor daily, usually over decades.
Picture: Safeguards during professional amalgam removal. Dental dam – gold nasal hood – Clean-Up suction tip.
Not shown: oxygen – nasal probe.
In the author’s practice the following protocol has been tested:
- Dental dam (rubber protection cloth – protection from splinters and fragments)
- Clean-Up suction tip (see above and additional protection from Hg vapor)
- Low-speed drill or extraction of the filling whole in order to avoid toxic mercury vapors
- Oxygenation via a nasal probe (oxidizes Hg, reducing absorption into the lung)
- Gold-coated nasal mask (gold intercepts mercury vapors), gold surgical mask for the dentist and his team
- Chlorella algae inlay in the cavity after amalgam removal (optional) – other options are possible
- Infusion with high-dose vitamin C and other micronutrients (optional) – reduces the immune response
Metal-free restorations
In biological dentistry the correct material plays a crucial role. Whether complex blood tests are done (LTT test) or whether materials are examined using bio-energy diagnostics (Autonomic Response Testing, Applied Kinesiology, Bioresonance, etc.) is up to the dentist. In an era of the hyperreactive, no longer tolerant immune system, the selection of the optimum substances should play an important role, before possibly mismatching or allergenic materials are permanently incorporated into the patient’s body. Because this could be the drop that makes the already brimming barrel overflow. It is also important to understand the entire body as an integrative system, instead of dividing it into its individual parts. In many cases it is common in the author’s practice to first shut down a primarily hyperreactive immune system through functionally medical aspects from the areas of nutritional-, environmental- and orthomolecular medicine, to remove all metals, inflammations and endodontically treated teeth and then to bring stability to this structure via long-term temporaries. Particularly with chronic progressive diseases such as MS, Parkinson’s, Alzheimer’s, cancer and ALS, this procedure is of crucial importance.
The material of choice, both for biocompatibility and aesthetics, is currently ceramic. Different types of ceramic are available to choose from: Constructions made of zirconia and more recently lithium disilicate ceramic (IPS e.max/ivoclar vivadent) have been well tested. Even the classic feldspar ceramic can be used.
The author is the Vice-President of the International Society of Metal Free Implantology (ISMI e.V.), and as such his clinical practice and all surgeries are performed free of metal.
Metal-free implants made of high-speed ceramic (zirconia) represent an important alternative to the usual titanium implants. According to Dr. Volker von Baehr (IMD Berlin), 15 to 20% of the population reacts incompatibly to titanium [79], mainly triggered by the massive use of titanium dioxide as filler or dye in medications, dietary supplements, hygiene products, cosmetics, chewing gum and toothpaste. Tissue-specific macrophages react to the titanium oxide particles primarily resulting from abrasion with phagocytosis and an increased nonspecific immune response (expression of “alarm cytokines” TNF-α, IL-1). Radar was able to show that zirconia particles of the same size in turn induce no pro-inflammatory cytokines (TNF-α) in macrophage cultures [80].
If one would also like to insert titanium for patients without risk, a titanium stimulation test should be done first in any case and a genetic predisposition to inflammation determined in the blood of patients with exaggerated immune response (high responder) to ensure that the patients will not be harmed by the implanted metal. However, the debate remains as to whether the metals work as an antenna in the mouth and thereby increase the patient’s electrical sensitivity.
Zirconia is a neutral element, translucent and biocompatible. Compared to titanium, zirconia does not possess free electrons on the surface, and therefore it cannot be in any way an interference field. Zirconia is also highly esthetic due to its white color in contrast to the grey of the titanium implants. With the help of the zirconia implant it is possible to combine biocompatibility and esthetics. Recently, zirconia implants have been available even as two-piece screw implants for all indications.
The biologically oriented dentist can also use PEEK (polyetheretherketone) screws for installation. This material has already long been used in orthopedic surgery as a replacement for intervertebral disks, since it is very similar to bone and cartilage in its elasticity. Both in Konstanz and in Tubingen, the author has used PEEK for some time for secondary frameworks, bridges or as the basis for one-piece cast prostheses. It is very flexible and can also be used from an osteopathic point of view for larger splinting, as it permits or can compensate for the independent movement of the sutures in contrast to the metal framework. Also, the chewing function benefits from this material, as its elasticity serves as a kind of shock absorber for chewing forces. This area is still new territory for dentistry; however, such innovations are needed to achieve long-term progress on the path to the optimum material; furthermore, the costs of a secondary framework made of PEEK are clearly lower than its metallic competitor.
All-ceramic materials have worked satisfactorily in the last few years not only in the areas of prosthetics and esthetics, but have for a long time also been applicable for surgical purposes. Also, the costs of the metal-free restorations were clearly reduced. In view of these facts, there is no longer any need to use base metal crowns in addition to supplies containing noble metals in the mouth or to screw gold abutments on titanium implants. Without exception the patient’s well-being should be paramount, which is possible in practically every situation with today’s existing materials and techniques.
Literature
- Richard Horton: The neglected epidemic of chronic disease. The Lancet, Volume 366, Issue 9496, Page 1514, 29 October 2005
- Strong K, Mathers C, Leeder S, Beaglehole R.: Preventing chronic diseases: how many lives can we save?The Lancet, Volume 366, Issue 9496, Pages 1578 – 1582, 29 October 2005
- Segade L, Suarez-Quintanilla D.: Otic ganglion parasympathetic neurons innervate the pulp of the mandibular incisor or the guinea pig.Neuroscience Letters 1988, 90(1-2) 33-38
- Ahlrot-Westerlund B: Mercury in cerebrospinal fluid in multiple sclerosis.Swed J Biol Med 1989, 1:6-7. 1. Richard Horton: The neglected epidemic of chronic disease. The Lancet, Volume 366, Issue 9496, Page 1514, 29 October 2005
- Strong K, Mathers C, Leeder S, Beaglehole R.: Preventing chronic diseases: how many lives can we save? The Lancet, Volume 366, Issue 9496, Pages 1578 – 1582, 29 October 2005
- Segade L, Suarez-Quintanilla D.: Otic ganglion parasympathetic neurons innervate the pulp of the mandibular incisor or the guinea pig. Neuroscience Letters 1988, 90(1-2) 33-38
- Ahlrot-Westerlund B: Mercury in cerebrospinal fluid in multiple sclerosis. Swed J Biol Med 1989, 1:6-7.
- Beck et. Al. : Oral disease, cardiovascular disease and systemic inflammation. Periodontology 2000; 23:110-20
- Ingalls T: Endemic clustering of multiple sclerosis in time and place, 1934-1984. Confirmation of a hypothesis. Am J Forensic Med Pathol 1986, 7:3-8.
- Meurman JH, Janket SJ, Qvarnström M, Nuutinen P.: Dental Infections and serum inflammatory markers in patients with and without severe heart disease, Oral Surg Oral Med Oral Path Oral Radiol endod 2003; 96:695-700.
- Mutter, Joachim: Is dental amalgam safe for humans? The opinion of the scientific committee of the European Commission. Journal of Occupational Medicine and Toxicology 2011, 6:2
- Perry VH, Newman TA, Cunningham C.: The impact of systemic infection on the progression of neurodegenerative disease. Nat Rev Neurosci. 2003 Feb;4(2):103-12.
- Stejskal J, Stejskal VD: The role of metals in autoimmunity and the link to neuroendocrinology. Neuro Endocrinol Lett 1999, 20:351-364.
- Siblerud RL: The relationship between mercury from dental amalgam and mental health. Am J Psychother 1989, 43:575-587.
- Siblerud RL, Motl J, Kienholz E: Psychometric evidence that mercury from silver dental fillings may be an etiological factor in depression, excessive anger, and anxiety. Psychol Rep 1994, 74:67-80.
- Wojcik DP, Godfrey ME, Haley B: Mercury toxicity presenting as chronic fatigue, memory impairment and depression: diagnosis, treatment, susceptibility, and outcomes in a New Zealand general practice setting (1994-2006). Neuro Endocrinol Lett 2006, 27:415-423.
- Leong CCW, Syed NI, Lorscheider FL: Retrograde degeneration of neurite membrane structural integrity of nerve growth cones following in vitro exposure to mercury. Neuro Report 2001, 12:733-737
- Stoiber T, Bonacker D, Bohm K: Disturbed microtubule function and induction of micronuclei by chelate complexes of mercury(II). Mutat Res 2004, 563:97-106.
- Stoiber T, Degen GH, Bolt HM, Unger E: Interaction of mercury(II) with the microtubule cytoskeleton in IMR-32 neuroblastoma cells. Toxicol Lett 2004, 151(Suppl 1):99-104.
- Thier R, Bonacker D, Stoiber T: Interaction of metal salts with cytoskeletal motor protein systems. Toxicol Lett 2003, 140:75-81.
- Pendergrass JC, Haley BE: Mercury-EDTA Complex Specifically Blocks Brain-Tubulin-GTP Interactions: Similarity to Observations in Alzheimer’s Disease. In Status Quo and Perspective of Amalgam and Other Dental Materials. International Symposium Proceedings. Edited by Friberg LT, Schrauzer GN. Stuttgart: Thieme Verlag; 1995:98-105.
- Pendergrass JC, Haley BE: Inhibition of brain tubulin-guanosine 5’-triphosphate interactions by mercury: similarity to observations in Alzheimer’s diseased brain. In MetalIons on Biological systems. Edited by Sigel A, Sigel H. New York: Dekker; 1997:461-478.
- Barregard J, Svalander C, Schutz A, Westberg G, Sällsten G, Blohmé I, Mölne J, Attman PO, Haglind P: Cadmium, mercury, and lead in kidney cortex of the general Swedish population: a study of biopsies from living kidney donors. Environ Health Perspect 1999, 107:867-871.
- Becker K, Kaus S, Krause C, Lepom P, Schulz C, Seiwert M, Seifert B: German Environmental Survey 1998 (GerES lll): environmental pollutants in blood of the German population. Int J Hyg Environ Health 2002, 205:297-308.
- Becker K, Schulz C, Kaus S, Seiwert M, Seifert B: German Environmental Survey 1998 (GerES III): Environmental pollutants in the urine of the German population. Int J Hyg Environ Health 2003, 206:15-24.
- Drasch G, Schupp I, Riedl G, Günther G: Einfluß von Amalgamfüllungen auf die Quecksilberkonzentration in menschlichen Organen. Dtsch Zahnärztl Z 1992, 47:490-496.
- Drasch G, Schupp I, Hofl H, Reinke R, Roider G: Mercury burden of human fetal and infant tissues. Eur J Ped 1994, 153:607-610.
- Drasch G, Wanghofer E, Roider G: Are blood, urine, hair, and muscle valid bio-monitoring parameters for the internal burden of men with the heavy metals mercury, lead and cadmium? Trace Elem Electrolyt 1997, 14:116-123.
- Eggleston DW, Nylander M: Correlation of dental amalgam with mercury in brain tissue. J Prosth Dent 1987, 58:704-707.
- Gottwald B, Traencker I, Kupfer J, Ganss C, Eis D, Schill WB, Gieler U: “Amalgam disease” — poisoning, allergy, or psychic disorder? Int J Hyg Environ Health 2001, 204:223-229.
- Guzzi G, Grandi M, Cattaneo C: Should amalgam fillings be removed? Lancet 2002, 360:2081.
- Guzzi G, Grandi M, Cattaneo C, Calza S, Minoia C, Ronchi A, Gatti A, Severi G: Dental amalgam and mercury levels in autopsy tissues: food for thought. Am J Forensic Med Pathol 2006, 27:42-45.
- Levy M, Schwartz S, Dijak M, Weber JP, Tardif R, Rouah F: Childhood urine mercury excretion: dental amalgam and fish consumption as exposure factors. Environ Res 2004, 94:283-290.
- Lorscheider FL, Vimy MJ, Summers AO: Mercury exposure from “silver” tooth fillings: emerging evidence questions a traditional dental paradigm. FASEB Journal 1995, 9:504-508.
- Kingman A, Albertini T, Brown LJ: Mercury concentrations in urine and whole blood associated with amalgam exposure in a US military population. J Dent Res 1998, 77:461-471.
- Mortada WI, Sobh MA, El-Defrawy MM, Farahat SE: Mercury in dental restoration: is there a risk of neph rotoxicity? J Nephrol 2002, 15:171-176.
- Nylander M: Mercury in pituitary glands of dentists. Lancet 1986, 22:442.
- Nylander M, Weiner J: Mercury and selenium concentrations and their interrelations in organs from dental staff and the general population. Br J Ind Med 1991, 48:729-734.
- Nylander M, Friberg L, Lind B: Mercury concentrations in the human brain and kidneys in relation to exposure from dental amalgam fillings. Swed Dent J 1987, 11:179-187.
- Pizzichini M, Fonzi M, Giannerini M, Mencarelli M, Gasparoni A, Rocchi G, Kaitsas V, Fonzi L: Influence of amalgam fillings on Hg levels and total antioxidant activity in plasma of healthy donors. Sci Total Environ 2003, 301:43-50.
- Weiner JA, Nylander M: The relationship between mercury concentration in human organs and different predictor variables. Sci Tot Environ 1993, 138:101-115.
- Zimmer H, Ludwig H, Bader M: Determination of mercury in blood, urine and saliva for the biological monitoring of an exposure from amalgam fillings in a group with self-reported adverse health effects. Int J Hyg Environ Health 2002, 205:205-211.
- Drasch G, Schupp I, Hofl H, Reinke R, Roider G: Mercury burden of human fetal and infant tissues. Eur J Ped 1994, 153:607-610.
- Ask K, Akesson A, Berglund M, Vahter M: Inorganic mercury and methylmercury in placentas of Swedish women. Environ Health Perspect 2002, 110:523-526.
- Holmes AS, Blaxill MF, Haley BE: Reduced levels of mercury in first baby haircuts of autistic children. Int J Toxicol 2003, 22:277-85.
- Morgan DL, Chanda SM, Price HC, Fernando R, Liu J, Brambila E, O’Connor RW, Beliles RP, Barone S Jr: Disposition of inhaled mercury vapor in pregnant rats: maternal toxicity and effects on developmental outcome. Toxicol Sci 2002, 66:261-273.
- Takahashi Y, Tsuruta S, Hasegawa J, Kameyama Y, Yoshida M: Release of mercury from dental amalgam fillings in pregnant rats and distribution of mercury in maternal and fetal tissues. Toxicology 2001, 163:115-126.
- Takahashi Y, Tsuruta S, Arimoto M, Tanaka H, Yoshida M: Placental transfer of mercury in pregnant rats which received dental amalgam restorations. Toxicology 2003, 185:23-33.
- Vahter M, Akesson A, Lind B, Bjors U, Schutz A, Berglund F: Longitudinal study of methylmercury and inorganic mercury in blood and urin of pregnant and lactating women, as well as in umbilical cord blood. Environ Res 2000, 84:186-194.
- Yoshida M, Satoh M, Shimada A, Yamamoto E, Yasutake A, Tohyama C: Maternal-to-fetus transfer of mercury in metallothionein-null pregnant mice after exposure to mercury vapor. Toxicology 2002, 175:215-222.
- Yoshida M, Watanabe C, Satoh M, Yasutake A, Sawada M, Ohtsuka Y, Akama Y, Tohyama C: Susceptibility of Metallothionein-Null Mice to the Behavioural Alterations Caused by Exposure to Mercury Vapour at Human-Relevant Concentration. Toxicol Sci 2004, 80:69-73.
- Drasch G, Aigner S, Roider G, Staiger F, Lipowskyn G: Mercury in human colostrum and early breast milk. Its dependence on dental amalgam and other factors. J Trace Elem Med Biol 1998, 12:23-27.
- Oskarsson A, Schultz A, Skerfving S, Hallen IP, Ohlin B, Lagerkvist BJ: Total and inorganic mercury in breast milk in relation to fish consumption and amalgam in lactating women. Arch Environ Health 1996, 51:234-241.
- Vimy MJ, Hooper DE, King WW, Lorscheider FL: Mercury from maternal “silver” tooth fillings in sheep and human breast milk. A source of neonatal exposure. Biol Trace Element Res 1997, 56:143-152.
- Hargreaves RJ, Evans JG, Janota I, Magos L, Cavanagh JB: Persistant mercury in nerve cells 16 years after metallic mercury poisoning. Neuropath Appl Neurobiol 1988, 14:443-452.
- Opitz H, Schweinsberg F, Grossmann T, Wendt-Gallitelli MF, Meyermann R: Demonstration of mercury in the human brain and other organs 17 years after metallic mercury exposure. Clin Neuropath 1996, 15:139-144.
- He F, Zhow X, Lin B, Xiung YP, Chen SY, Zhang SL, Ru JY, Deng MH: Prognosis of Mercury poisoning in mercury refinery workers. Ann Acad Med Singapore 1984, 13:389-393.
- Kishi R, Doi R, Fukushi Y, Satoh H, Ono A: Residual neurobehavioural effects associated with chronic exposure to mercury vapour. Occup Environ Med 1994, 51:35-41.
- Kobal A, Horvat M, Prezelj M, Briski AS, Krsnik M, Dizdarevic T, Mazej D, Falnoga I, Stibilj V, Arneric N, Kobal D, Osredkar J: The impact of long-term past exposure to elemental mercury on antioxidative capacity and lipid peroxidation in mercury miners. J Trace Elem Med Biol 2004, 17:261-274.
- Letz R, Gerr F, Cragle D, Green R, Watkins J, Fidler A: Residual neurologic deficits 30 years after occupational exposure to elemental mercury. Neurotoxicology 2000, 21:459-474.
- Sugita M: The biological half-time of heavy metals. The existence of a third, `slowest’ component. Int Arch Occup Environ Health 1978, 41:25-40.
- Takahata N, Hayashi H, Watanabe S, Anso T: Accumulation of mercury in the brains of two autopsy cases with chronic inorganic mercury poisoning. Folia Psychiatr Neurol Jpn 1970, 24:59-69.
- Lindh U, Hudecek R, Dandersund A, Eriksson S, Lindvall A: Removal of dental amalgam and other metal alloys supported by antioxidant therapy alleviates symptoms and improves quality of life in patients with amalgam-associated ill health. Neuro Endocrinol Lett 2002, 23:459-482.
- Siblerud RL: A comparison of mental health of multiple sclerosis patients with silver/mercury dental fillings and those with fillings removed. Psychol Rep 1992, 70:1139-1151.
- Huggins HA, Levy TE: Cerebrospinal fluid protein changes in multiple sclerosis after dental amalgam removal. Altern Med Rev 1998, 4:295-300.
- Bates M, Fawcett J, Garrett N, Cutress T, Kjellstrom T: Related articles, health effects of dental amalgam exposure: a retrospective cohort study. Int J Epidemiol 2004, 33:894-902.
- Engel P: Beobachtungen über die Gesundheit vor und nach Amalgamentfernung. [Observations on health before and after removing dental amalgam]. Schweiz Monatsschr Zahnm 1998, 108:2-14.
- Heintze U, Edwardsson S, Derand T, Birkhed D: Methylation of mercury from dental amalgam and mercuric chloride by oral streptococci in vitro. Scand J Dent Re 1983, 91:150-152.
- Leistevuo J, Leistevuo T, Helenius H, Pyy L, Osterblad M, Huovinen P, Tenovuo J: Dental amalgam fillings and the amount of organic mercury in human saliva. Caries Res 2001, 35:163-166.
- Yannai S, Berdicevsky I, Duek L: Transformations of inorganic mercury by Candida albicans and Saccharomyces cerevisiae. Appl Environ Microbiol 1991, 57:245-247.
- Harris HH, Pickering IJ, George GN: The chemical form of mercury in fish. Science 2003, 301:1203.
- Claesson et al.: Production of volatile sulfur compounds by various Fusobacterium species. Oral Microbiol. Immunol. 1990; 5:137-142
- Langendijk PS, Hanssen JT, Van der Hoeven JS.: Sulfate-reducing bacteria in association with human periodontitis. J Clin Periodontol Dec 2000;27(12):943-50.
- Persson S., Edlund MB., Claesson, R., Carlsson J.: The Formation of hydrogen sulfide and methyl mercaptan by oral bacteria. Oral Microbiology and Immunology 1990 August; Vol. 5 (4): 195-201.
- Lechner J.: Immunstress durch Zahnmetalle und Elektrosmog. Raum&Zeit 1995, 74: 5-13.
- Virtanen H, Huttunen J, Toropainen A, Lappalainen R.: Interaction of mobile phones with superficial passive metallic implants. Phys Med Biol. 2005 Jun 7;50(11):2689-700.
- Klinghardt D: Neural Therapy & Mesotherapy Course A: The Intensive. Klinghardt Academy 2011, 80-82.
- Nischwitz D: Die Wurzel allen Übels. Endo Tribune, in Dental Tribune 5/14: 21. Online: http://www.zwp-online.info/de/fachgebiete/implantologie/grundlagen/die-wurzel-allen-uebels
- Lechner J.: Kavitätenbildende Osteolysen des Kieferknochens. 1. Auflage April 2011, München.
- Price WA: Nutrition and Physical Degeneration (1939-2003). 6. vollst. überarb. u. erw. Auflage. The Price-Pottenger Nutrition Foundation, Inc., La Mesa, CA.
- Straub RH, Cutolo M, Buttgereit F, Pongratz G.: Energy regulation and neuroendocrine-immune control in chronic inflammatory diseases. J Intern Med. 2010 Jun;267(6):543-60.
- Schütt S, Von Baehr V.: Hyperreeaktivität von Gewebemakrophagen nach Kontakt mit Titanoxidpartikeln als Ursache einer verstärkten lokalen Entzündungsreaktion bei Patienten mit Periimplantitis. ZWR – Das
Deutsche Zahnärzteblatt 2010, 119: 222-232
- Radar CP, Sterner T, Jakob F et al.: Cytokine response of human macrophage-like cells after contact with polyethylene and pure titanium particles. J Arthroplasty 1999; 14: 840-848